Two oral presentations and multiple posters on clinical and
preclinical-stage candidates to be presented at SITC, demonstrating
the strength of Immatics’ TCR-T PRAME franchise to target solid cancers
- ACTengine® IMA203 demonstrates 54% cORR, 12.1 months mDOR and 6 months mPFS in heavily pretreated metastatic melanoma patients and >1-year mPFS in patients with deep responses; Company plans to start its randomized-controlled Phase 3 SUPRAME trial in December 2024 to evaluate IMA203 in second-line or later metastatic melanoma
- Next-generation ACTengine® IMA203CD8 TCR-T cell therapy targeting PRAME demonstrates enhanced pharmacology and potency per cell; Phase 1a dose escalation reinitiated to target higher doses, positioning this TCR-T candidate for future development in solid cancers with medium-level PRAME copy numbers, such as ovarian cancer, endometrial cancers and triple-negative breast cancer
- A first update on Immatics’ Bispecific TCER® IMA402 targeting PRAME and initial clinical data from the ongoing Phase 1a dose escalation trial is expected to be reported by year-end
Houston, Texas and Tuebingen, Germany, November 8, 2024 – Immatics N.V. (NASDAQ: IMTX, “Immatics” or the “Company”), a clinical-stage biopharmaceutical company active in the discovery and development of T cell-redirecting cancer immunotherapies, today announced an expanded clinical dataset from the ongoing Phase 1b dose expansion clinical trial for ACTengine® IMA203 in addition to updated Phase 1 dose escalation clinical data on its next-generation ACTengine® IMA203CD8 TCR-T cell therapy. For the first time, the Company also reported preclinical data on other next-generation T cell candidates and combination strategies as part of its strategy to further exploit opportunities in additional solid tumor types within its PRAME franchise.
All dates and times of Immatics’ upcoming oral and poster presentations at the 39th Annual Meeting of the Society for Immunotherapy of Cancer (SITC) are available here. The data slides are accessible in the ‘Events & Presentations’ section of the Investor & Media section of the Company’s website.
“Immatics remains fully focused on the clinical development of our most advanced lead product candidate, IMA203, in second-line or later metastatic melanoma patients. We look forward to the initiation of SUPRAME, the registration-enabling Phase 3 trial, in December,” said Dr. Cedrik Britten, Chief Medical Officer at Immatics. “Today, we also provide an update on our first, next-generation cell therapy, IMA203CD8, which is designed to achieve enhanced anti-tumor activity. The data announced confirm IMA203CD8’s enhanced pharmacology and potency per cell in patients. These attributes highlight the potential of this therapy in hard-to-treat solid tumors with medium-level PRAME copy numbers, including ovarian, endometrial and triple-negative breast cancer. The next step will be to further increase the cell dose to assess the full clinical potential of IMA203CD8 beyond melanoma. In addition, we strive to continuously improve the potential therapeutic benefit for patients with a range of PRAME-positive cancers through the expansion of our PRAME franchise.”
ACTengine® IMA203 Monotherapy Phase 1b Trial - Clinical Data and Development Path Summary
On October 10, 2024, Immatics provided a data update on IMA203 monotherapy in 281 heavily pretreated metastatic melanoma patients from the ongoing Phase 1b dose expansion part of the clinical trial in which patients were treated at the recommended Phase 2 dose (RP2D, 1 to 10 billion total TCR-T cells).
The data announced today include all infused patients in the Phase 1b dose expansion part of the trial (N=412), consisting of the 28 melanoma patients reported on October 10, 2024, and 13 non-melanoma patients, of which 10 non-melanoma patients were reported on November 8, 2023.
IMA203 monotherapy has maintained a favorable tolerability profile with no treatment-related Grade 5 events in the entire safety population (N=703 Phase 1a and Phase 1b patients across all dose levels and all tumor types).
Best Overall Response for IMA203 in Dose Expansion in All Indications (N=41#)
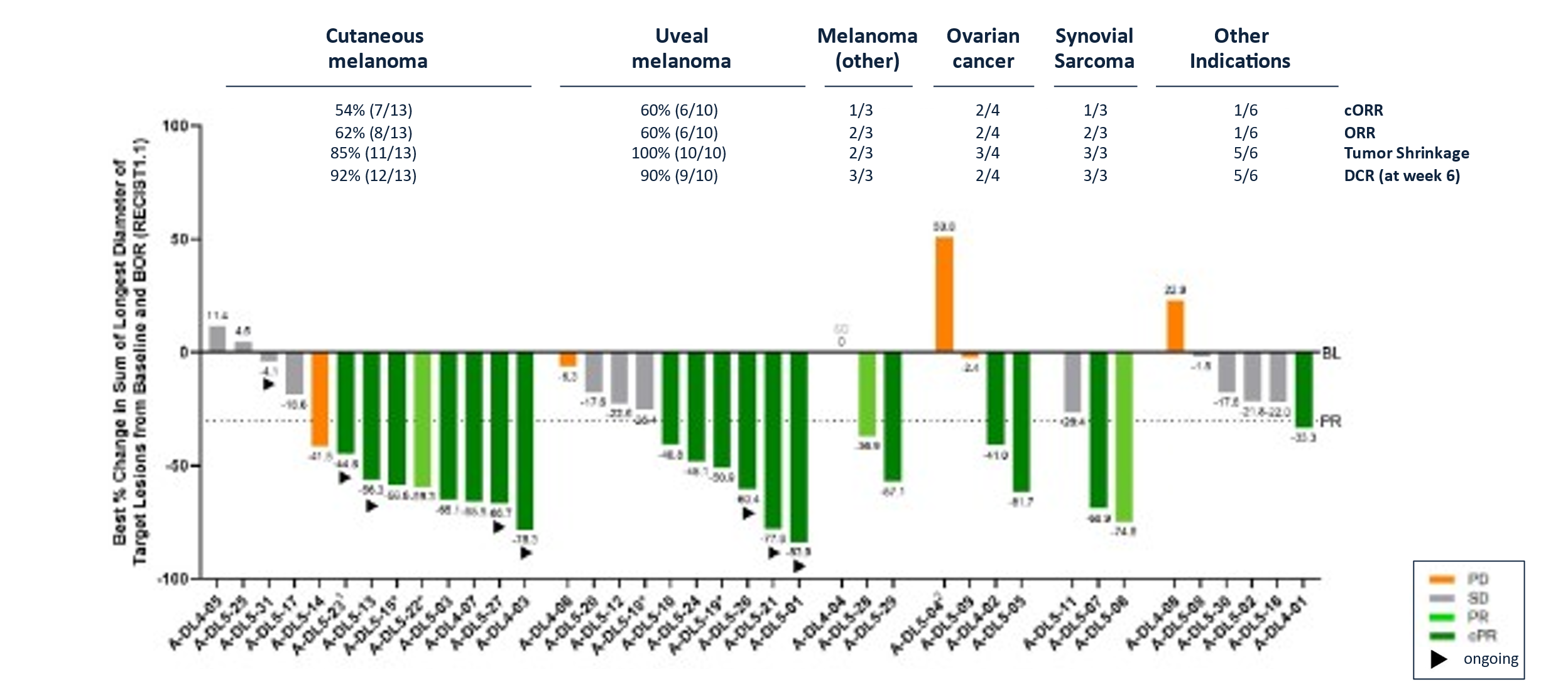
Data cut-off Aug 23, 2024; #First tumor assessment post infusion pending for 2/28 melanoma patients at data-cut; *Maximum change of target lesions and RECIST1.1 response at different timepoints. 1Patient A-DL5-23 is off study at data cut-off; 2Patient received one dose nivolumab erroneously.
Development Path for IMA203
Based on the Phase 1b data, the Company is on track to commence SUPRAME, the registration-enabling Phase 3 randomized-controlled clinical trial in melanoma for IMA203, in December 2024.
SUPRAME will evaluate IMA203 targeting PRAME in 360 HLA-A*02:01-positive patients with second-line or later (2L+) unresectable or metastatic melanoma who have received prior treatment with a checkpoint inhibitor. Patients will be randomized 1:1 for IMA203 or investigator’s choice of selected approved treatments in the 2L+ setting, including nivolumab/relatlimab, nivolumab, ipilimumab, pembrolizumab, lifileucel (US only) or chemotherapy. The primary endpoint for full approval will be median PFS and secondary endpoints will include objective response rate, safety, duration of response, no overall survival detriment and patient-reported outcomes.
Patient enrollment for SUPRAME is forecast to be completed in 2026, and a pre-specified interim analysis is planned for early 2026. Immatics aims to submit a Biologics License Application (BLA) in early 2027 for full approval.
ACTengine® IMA203CD8 (GEN2) Monotherapy Phase 1 Dose Escalation Trial - Patient Population & Clinical Data Summary
Patient population: Heavily pretreated patients with solid tumors
As of data cut-off on September 30, 2024, 444 heavily pretreated HLA-A*02:01 and PRAME-positive patients with solid tumors were infused with IMA203CD8 monotherapy across four escalating dose levels, of which 415 patients were evaluable for efficacy. The median total infused dose was 1.48x109 TCR-T cells, and the patient population is composed of patients with a median of three lines of prior systemic treatments.
Safety: Treatment with IMA203CD8 demonstrates a manageable tolerability profile across dose levels
IMA203CD8 monotherapy has maintained a manageable tolerability profile in the 44 patients treated. The most frequent adverse events at or above Grade 3 were expected cytopenia associated with lymphodepletion. Some patients also experienced mild to moderate CRS (Grade 1: 36% Grade 2: 48% Grade 3: 11% Grade 4: 2%).
As previously reported, two patients experienced dose-limiting toxicities at dose level 4b, which prompted a dosing adjustment to dose level 4a. After further assessing the tolerability profile of IMA203CD8 in additional patients treated at dose level 4a, the eligibility criteria and the IL-2 dose regimen were modified, and dose escalation beyond dose level 4a was reinitiated. One Grade 5 adverse event classified as possibly related to treatment with IMA203CD8 was also observed as reported previously in March 2024. The maximum tolerated dose has not yet been determined.
Anti-tumor activity and durability: Deep and durable objective responses observed
- As of data cut-off on September 30, 2024, 10 of 17 responses were ongoing, of which three confirmed responses were ongoing at 14+, 15+ and 24+ months.
- Of note, these patients had been treated at substantially lower doses compared to IMA203 (GEN1), i.e. in a range of 0.2-0.48x109 TCR-T cells/m2 BSA (dose level 3) to 0.801-1.2x109 TCR-T cells/m2 BSA (dose level 4c) T cells infused.
- Deep responses with ≥50% tumor size reduction were observed in 11 out of 17 responders. This group included two patients with complete response of target lesions, of which one patient showed a complete metabolic response according to PET-CT scan6.
- 41% (14/34) confirmed objective response rate (cORR) and 41% (17/41) objective response rate (ORR).
- Median duration of response (mDOR) of 9.2 months at a median follow-up (mFU) of 13.1 months.
- Tumor shrinkage7 of 84% (32/38) and disease control rate8 at week 6 of 85% (34/40).
Translational data: Opportunity of IMA203CD8 in medium-level PRAME expressing indications
Translational data indicate that PRAME expression level is associated with clinical activity in IMA203 and IMA203CD8 treated patients. Both IMA203 and IMA203CD8 achieved deep responses despite IMA203CD8 patients receiving lower product doses. Based on the enhanced pharmacology of IMA203CD8, the evaluation of higher doses of IMA203CD8 in the ongoing dose escalation trial opens the possibility of addressing hard-to-treat solid tumor indications with a medium-level of PRAME copy numbers, such as ovarian cancer, endometrial cancers and triple-negative breast cancer.
Preclinical Data on New Approaches for TCR-T Based Cell Therapies
As part of Immatics’ long-term strategy to expand its PRAME franchise, the Company has conducted preclinical studies for the potential future clinical development of next-generation TCR-T-based cell therapies targeting PRAME to further enhance the efficacy and durability of IMA203. These efforts include the evaluation of TCR-T cells armored with membrane-bound IL-15 (mbIL15) targeting tumor types with low PRAME copy numbers, such as squamous non-small-cell lung cancer and squamous head and neck cancers. In addition, the Company is developing an allogeneic cell therapy approach to further increase commercial attractiveness and to reach patients quickly with its next-generation off-the-shelf cell therapy, ACTallo®. The preclinical data will be presented during poster sessions at SITC.
About ACTengine® IMA203, IMA203CD8 and Target PRAME
ACTengine® IMA203 is Immatics’ most advanced TCR-based autologous cell therapy that is directed against an HLA-A*02-presented (human leukocyte antigen) peptide derived from preferentially expressed antigen in melanoma (PRAME), a protein frequently expressed in a large variety of solid cancers. PRAME is homogeneously and specifically expressed in tumor tissue and Immatics’ PRAME peptide is present at a high copy number per tumor cell. The peptide has been identified and characterized by Immatics’ proprietary mass spectrometry-based target discovery platform, XPRESIDENT®. Through its proprietary TCR discovery and engineering platform XCEPTOR®, Immatics has generated a highly specific T cell receptor (TCR) against this target for ACTengine® IMA203.
ACTengine® IMA203 TCR-T is currently being evaluated as a monotherapy in a Phase 1 clinical trial in patients with solid tumors expressing PRAME, such as cutaneous melanoma. An IMA203 registration-enabling randomized controlled Phase 3 trial, “SUPRAME,” is planned to commence in December 2024.
ACTengine® IMA203 TCR-T is also currently being evaluated in Phase 1 IMA203CD8 (GEN2) monotherapy, where IMA203 engineered T cells are co-transduced with a CD8αβ co-receptor.
- END -
About Immatics
Immatics combines the discovery of true targets for cancer immunotherapies with the development of the right T cell receptors with the goal of enabling a robust and specific T cell response against these targets. This deep know-how is the foundation for our pipeline of Adoptive Cell Therapies and TCR Bispecifics as well as our partnerships with global leaders in the pharmaceutical industry. We are committed to delivering the power of T cells and to unlocking new avenues for patients in their fight against cancer.
Immatics intends to use its website www.immatics.com as a means of disclosing material non-public information. For regular updates you can also follow us on X, Instagram and LinkedIn.
Forward-Looking Statements
Certain statements in this press release may be considered forward-looking statements. Forward-looking statements generally relate to future events or the Company’s future financial or operating performance. For example, statements concerning timing of data read-outs for product candidates, the timing, outcome and design of clinical trials, the nature of clinical trials (including whether such clinical trials will be registration-enabling), the timing of IND or CTA filing for pre-clinical stage product candidates, the timing of BLA filings for clinical stage product candidates, estimated market opportunities of product candidates, the Company’s focus on partnerships to advance its strategy, and other metrics are forward-looking statements. In some cases, you can identify forward-looking statements by terminology such as “may”, “should”, “expect”, “plan”, “target”, “intend”, “will”, “estimate”, “anticipate”, “believe”, “predict”, “potential” or “continue”, or the negatives of these terms or variations of them or similar terminology. Such forward-looking statements are subject to risks, uncertainties, and other factors which could cause actual results to differ materially from those expressed or implied by such forward-looking statements. These forward-looking statements are based upon estimates and assumptions that, while considered reasonable by Immatics and its management, are inherently uncertain. New risks and uncertainties may emerge from time to time, and it is not possible to predict all risks and uncertainties. Factors that may cause actual results to differ materially from current expectations include, but are not limited to, various factors beyond management's control including general economic conditions and other risks, uncertainties and factors set forth in the Company’s Annual Report on Form 20-F and other filings with the Securities and Exchange Commission (SEC). Nothing in this press release should be regarded as a representation by any person that the forward-looking statements set forth herein will be achieved or that any of the contemplated results of such forward-looking statements will be achieved. You should not place undue reliance on forward-looking statements, which speak only as of the date they are made. The Company undertakes no duty to update these forward-looking statements. All the scientific and clinical data presented within this press release are – by definition prior to completion of the clinical trial and a clinical study report – preliminary in nature and subject to further quality checks including customary source data verification.
For more information, please contact:
| Media | |
| Trophic Communications | |
| Phone: +49 171 3512733 | |
| immatics@trophic.eu |
| Immatics N.V. | |
| Jordan Silverstein | |
| Head of Strategy | |
| Phone: +1 346 319-3325 | |
| InvestorRelations@immatics.com |
1 Includes one patient who started lymphodepletion but did not receive IMA203 TCR-T cells.
2 All infused patients, first tumor assessment post infusion pending for 2/28 melanoma patients at data-cut.
3 All patients who started lymphodepletion as of the data cut-off on August 23, 2024.
4 All patients who started lymphodepletion.
5 All infused patients with at least one tumor assessment postbaseline.
6 Metabolic CR on investigator-initiated PET month 14 post infusion.
7 Three patients excluded from tumor shrinkage analysis and figures due to lack of post-treatment assessment.
8 One patient had an early tumor assessment, outside the first assessment visit window and is not included in DCR calculation.
Attachment















